Sophmore Chemistry Classwork
Charmemes
The first activity we did this year was to create a meme, a "Charmeme".
A Charmeme is a funny Chemistry Cat meme that incorporates a section of either the syllabus or computer policy. The goal is to teach everyone about a serious topic in a humorous way. To create a Charmeme, we first looked over the class syllabus and the computer policy and found something we found important. In my case my partner and used a passage from the computer usage policy, in the other things to consider section:
"Share Ideas thoughts and feelings--not passwords"
We then set about to take that passage and make a funny meme out of it. We reverted to periodic table related humor. The Charmeme is attached below.
A Charmeme is a funny Chemistry Cat meme that incorporates a section of either the syllabus or computer policy. The goal is to teach everyone about a serious topic in a humorous way. To create a Charmeme, we first looked over the class syllabus and the computer policy and found something we found important. In my case my partner and used a passage from the computer usage policy, in the other things to consider section:
"Share Ideas thoughts and feelings--not passwords"
We then set about to take that passage and make a funny meme out of it. We reverted to periodic table related humor. The Charmeme is attached below.
Bonding Basics: Ionic Bonds
In this activity I learned how to write/draw elements and their ionic bonds using Lewis structures. Lewis structures are visual representations of elements in which there is the element symbol is surrounded by dots that represent that element's valence electrons. The valence electron dots have to be drawn in a specific way. Imagine the element symbol in a box. First draw a dot at the top left of the box, then at the top of the right wall, then at the right of the bottom wall, etc.
I was given a worksheet with several element bonds written out. For example, Sodium+Chlorine. I then Wrote each element individually using Lewis structures. In Sodium and Chlorine's case: Na and Cl
After that I drew an arrow from the electrons of the element losing electrons (Sodium, because it has less valence electrons) to the missing spaces of the element receiving them (Chlorine, because it has almost all of its valence electrons), so that both can become stable. Afterwards I drew the lewis structure for the combined elements, or the compound. Finally, I wrote the formula for the compound: NaCl
Sometimes, more that one of the same element was required in order to completely fill the electron shell of the other. In this case, I simply added another one of that element's Lewis structure and wrote how many of that element was needed in order to create the bond. For example, for water, which needs 2 hydrogen atoms to fill oxygen's valence electron shell I wrote H2O. In this activity I revisited what an ionic bond was as well as learned how to draw ionic bonds using Lewis structures. This was an important activity because Lewis structures are commonly used to represent elements and their bonds as a result of their neatness. It is important to understand how they work and how to use them. Ionic bonds are also one of the three ways elements can bond with each other. In Ionic Bonding, one atom takes a valence electron from another atom in order to complete it's electron shell and become stable.
I was given a worksheet with several element bonds written out. For example, Sodium+Chlorine. I then Wrote each element individually using Lewis structures. In Sodium and Chlorine's case: Na and Cl
After that I drew an arrow from the electrons of the element losing electrons (Sodium, because it has less valence electrons) to the missing spaces of the element receiving them (Chlorine, because it has almost all of its valence electrons), so that both can become stable. Afterwards I drew the lewis structure for the combined elements, or the compound. Finally, I wrote the formula for the compound: NaCl
Sometimes, more that one of the same element was required in order to completely fill the electron shell of the other. In this case, I simply added another one of that element's Lewis structure and wrote how many of that element was needed in order to create the bond. For example, for water, which needs 2 hydrogen atoms to fill oxygen's valence electron shell I wrote H2O. In this activity I revisited what an ionic bond was as well as learned how to draw ionic bonds using Lewis structures. This was an important activity because Lewis structures are commonly used to represent elements and their bonds as a result of their neatness. It is important to understand how they work and how to use them. Ionic bonds are also one of the three ways elements can bond with each other. In Ionic Bonding, one atom takes a valence electron from another atom in order to complete it's electron shell and become stable.
Periodic Quest
Methods
------------
1) Group together the elements based on how they react with chemicals.
Group 1 Group 2 Group 3 Group 4
(no reaction) (C1 & C2) (C2 & C4) (C3)
Ar, Ta & Cn Cp, Sa, Sc Pi, Ge, Lb Le, Aq
2) You should have 4 groups of elements. Organize the elements into a chart of 3 rows and 4 columns. You will have one empty space.
No reaction React w/ C1 & C2 React w/ C2 & C4 React w/ C3
Ar Cp Pi Le
Ta Sa Ge Aq
Cn Sc Lb
3) How do you think the elements should be organized by mass? Make sure the elements are organized by mass in a way that makes sense.
The elements should be organized by mass in such a way that the masses of the elements go from lowest to highest from left to right as well as from top to bottom. However, the columns already in place that group the elements by reactivity should remain the same. Therefore, the elements can be rearranged in their reactivity column but not moved out of it.
4) Copy your chart onto a piece of scratch paper, numbering your elements from one (upper left) to twelve (bottom right). Remember, there will be ONE missing element.
React w/ C1 & C2 React w/ C2 & C4 React w/ C3 React w/None
1. Cp Mass:3.1 2. Pi Mass:6.2 3. Aq Mass:9.4 4. Ar Mass:11.8
5. Sc Mass:14.1 6. Ge Mass:16.4 7. 8. Ta Mass:20.9
9. Sa Mass:25.1 10. Lb Mass:27.2 11. Le Mass:29.1 12. Cn Mass:32.3
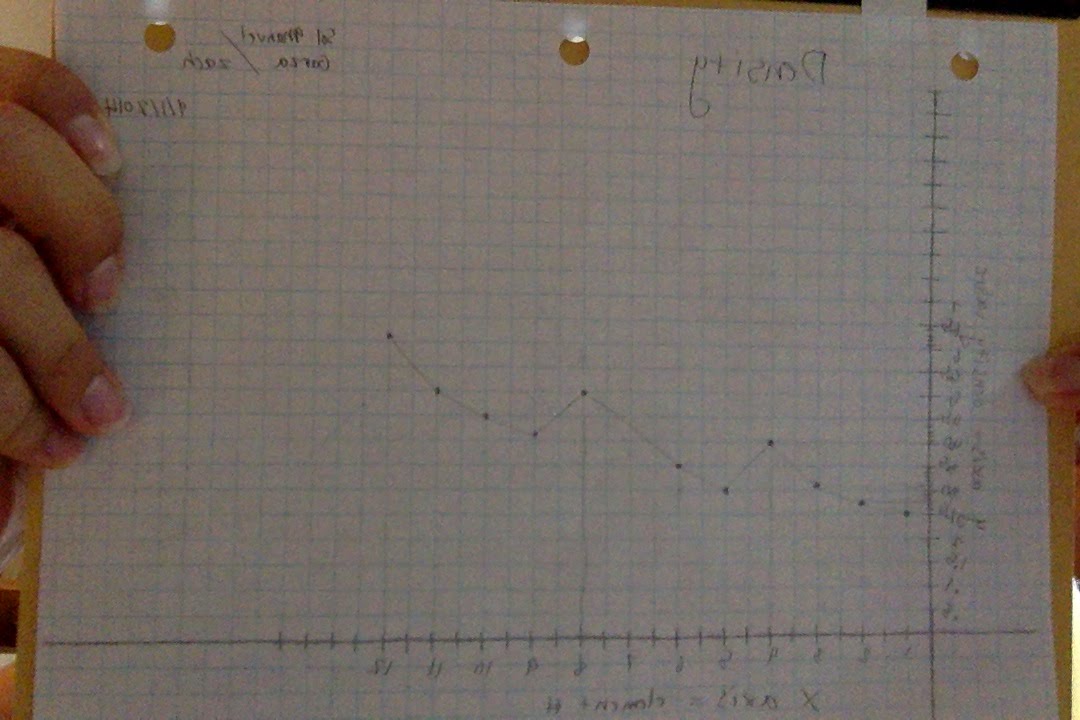
5) Using graph paper plot the density values of each element. Density value belongs on the y axis and the element number belongs on the x axis. Connect the points.
Density Graph:
------------
1) Group together the elements based on how they react with chemicals.
Group 1 Group 2 Group 3 Group 4
(no reaction) (C1 & C2) (C2 & C4) (C3)
Ar, Ta & Cn Cp, Sa, Sc Pi, Ge, Lb Le, Aq
2) You should have 4 groups of elements. Organize the elements into a chart of 3 rows and 4 columns. You will have one empty space.
No reaction React w/ C1 & C2 React w/ C2 & C4 React w/ C3
Ar Cp Pi Le
Ta Sa Ge Aq
Cn Sc Lb
3) How do you think the elements should be organized by mass? Make sure the elements are organized by mass in a way that makes sense.
The elements should be organized by mass in such a way that the masses of the elements go from lowest to highest from left to right as well as from top to bottom. However, the columns already in place that group the elements by reactivity should remain the same. Therefore, the elements can be rearranged in their reactivity column but not moved out of it.
4) Copy your chart onto a piece of scratch paper, numbering your elements from one (upper left) to twelve (bottom right). Remember, there will be ONE missing element.
React w/ C1 & C2 React w/ C2 & C4 React w/ C3 React w/None
1. Cp Mass:3.1 2. Pi Mass:6.2 3. Aq Mass:9.4 4. Ar Mass:11.8
5. Sc Mass:14.1 6. Ge Mass:16.4 7. 8. Ta Mass:20.9
9. Sa Mass:25.1 10. Lb Mass:27.2 11. Le Mass:29.1 12. Cn Mass:32.3
5) Using graph paper plot the density values of each element. Density value belongs on the y axis and the element number belongs on the x axis. Connect the points.
Density Graph:
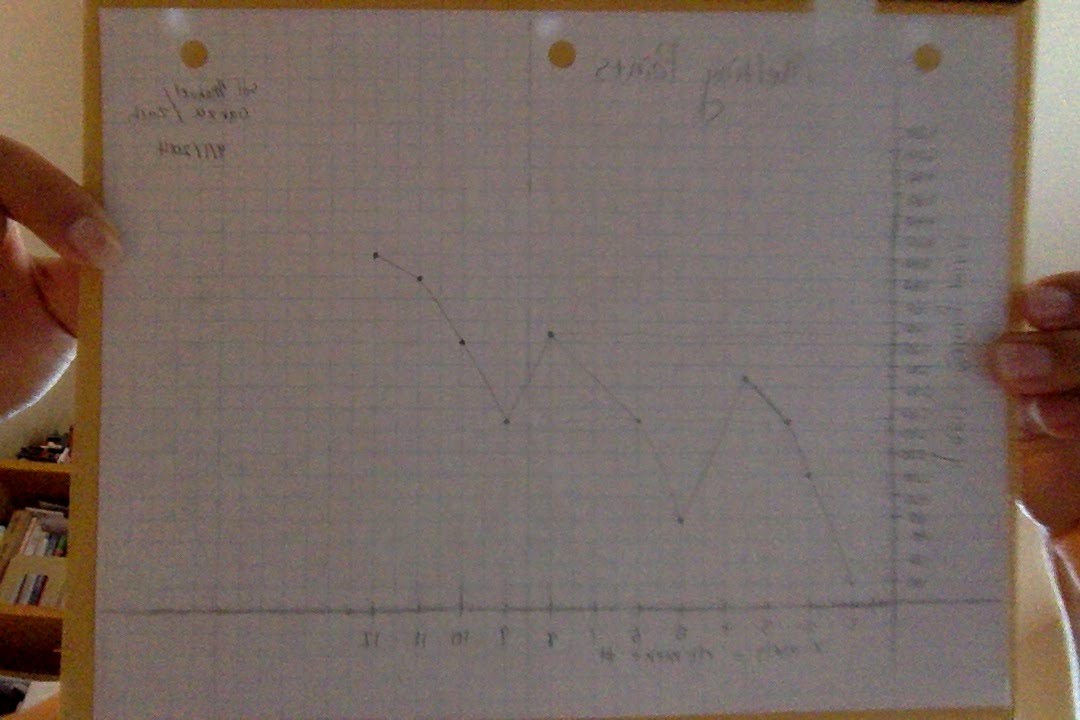
6) Plot a similar graph using the melting points of all the elements.
Melting point Graph:
Melting point Graph:
Discussion
------------------1) Describe the pattern of your density graph and your melting point graph.
The density and melting point graphs both seemed to steadily climb one or two units at a time as the element number got higher. However, every time a new row was started, the density and melting point fell about two units. This happened for both graphs. Essentially, the graphs would go up four spaces and then down one.
2) Predict the density and melting point of the missing element.
I predict that the melting point will be 290. I believe this because it is where the line is on the graph. The density would be 3.7. This is because it follows the pattern that emerges on the graph.
3) Can you predict the atomic mass of the missing element?
Because the second row in the table that organizes the elements by atomic mass has escalating masses that increase by approximately 2.3, I predicted that the missing element would have an atomic mass of 18.6. This is because 16.4+2.2= 18.6 and 18.6+2.3=20.9. It seems to be the number that fits best.
4) What colour do you expect the missing element to be? Why?
The missing element is in the column of elements that only react with C3. All the elements that react with C3 are orange. Therefore, I believe that because the missing element is in that column, it will be orange.
5) How do you expect that the missing element will react with chemicals C1, C2, C3 and C4?
Because the missing element is in the column of elements that only reacted with chemical C3, I believe that the mystery element will also only react to C3.
6) Select a Name for your new element and create a symbol.
The element will be named Solzachium and will be represented by the symbol Sz.
Challenge Questions
----------------------------------
1) Draw your new periodic table. Your table should include element name, element symbol and atomic number.
React w/ C1 & C2 React w/ C2 & C4 React w/ C3 React w/None
1. Cp Mass:3.1 2. Pi Mass:6.2 3. Aq Mass:9.4 4. Ar Mass:11.8
5. Sc Mass:14.1 6. Ge Mass:16.4 7. 8. Ta Mass:20.9
9. Sa Mass:25.1 10. Lb Mass:27.2 11. Le Mass:29.1 12. Cn Mass:32.3
------------------1) Describe the pattern of your density graph and your melting point graph.
The density and melting point graphs both seemed to steadily climb one or two units at a time as the element number got higher. However, every time a new row was started, the density and melting point fell about two units. This happened for both graphs. Essentially, the graphs would go up four spaces and then down one.
2) Predict the density and melting point of the missing element.
I predict that the melting point will be 290. I believe this because it is where the line is on the graph. The density would be 3.7. This is because it follows the pattern that emerges on the graph.
3) Can you predict the atomic mass of the missing element?
Because the second row in the table that organizes the elements by atomic mass has escalating masses that increase by approximately 2.3, I predicted that the missing element would have an atomic mass of 18.6. This is because 16.4+2.2= 18.6 and 18.6+2.3=20.9. It seems to be the number that fits best.
4) What colour do you expect the missing element to be? Why?
The missing element is in the column of elements that only react with C3. All the elements that react with C3 are orange. Therefore, I believe that because the missing element is in that column, it will be orange.
5) How do you expect that the missing element will react with chemicals C1, C2, C3 and C4?
Because the missing element is in the column of elements that only reacted with chemical C3, I believe that the mystery element will also only react to C3.
6) Select a Name for your new element and create a symbol.
The element will be named Solzachium and will be represented by the symbol Sz.
Challenge Questions
----------------------------------
1) Draw your new periodic table. Your table should include element name, element symbol and atomic number.
React w/ C1 & C2 React w/ C2 & C4 React w/ C3 React w/None
1. Cp Mass:3.1 2. Pi Mass:6.2 3. Aq Mass:9.4 4. Ar Mass:11.8
5. Sc Mass:14.1 6. Ge Mass:16.4 7. 8. Ta Mass:20.9
9. Sa Mass:25.1 10. Lb Mass:27.2 11. Le Mass:29.1 12. Cn Mass:32.3